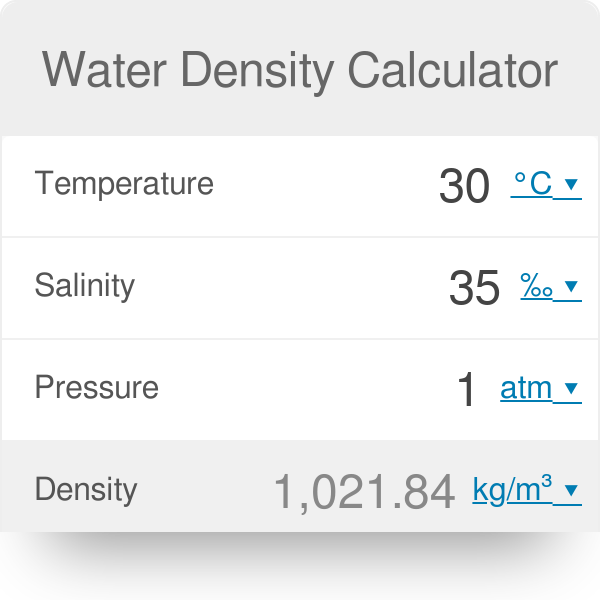
These electrostatic effect give rise to a shrinkage of the water. In this way the field tends to disrupt hydrogen bonded structures in liquid water, and to compress the water molecules surrounding an ion. The interaction of the electrostatic field of an ion with water tends to align the dipolar water molecules in the direction of the field. Density of ocean water at the sea surface is about 1027 kg/m 3. Ocean water is more dense because of the salt in it. 
The density of pure water is 1000 kg/m 3. 
This is applicable for any fluid that is primarily water based. See this excerpt from a study on the volumetric effects due to ion-solvent interaction in aqueous electrolyte solutions: Then click on the Calculate button and the water density is calculated and displayed, based on your input data. In this more ordered arrangement, the ions effectively fill the voids between the water molecules, and the volume of the water only increases slightly. The positive Hydrogen ends of the water molecules are attracted to the negatively charged Cl ions, and the positive oxygen ends are attracted to the positively charged Na ions. Because of the geometry of water molecules, they are essentially dipoles with “positive and a negative ends”. Interestingly, the dissolved salt does not increase the volume of the water by the volume of the added salt, and this is due to the charge of the Na and Cl ions and the H 2O molecules. The output dynamic viscosity is given as cP, mPas, Pas, Ns/m / (fth), while the kinematic viscosity is given as cSt, m /s, and ft. When salt (sodium chloride or NaCl) dissolves in water, there is a significant increase in mass of the solution due to the relatively higher molecular mass of the dissolved ions Na (22 g/mol) and Cl (35.5g/mol) when compared to water or H 2O (20 g/mol). The calculator below can be used to calculate the liquid water dynamic or kinematic viscosity at given temperatures.
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
